Diabetic Mouse Delayed Wound Model Following Treatment with the NerveStim™ Neuropathy System
Abstract
Delayed wound healing in diabetes is characterized by impaired angiogenesis, persistent inflammation, extracellular matrix dysregulation, and peripheral neuropathy. A preclinical study was conducted using a diabetic mouse delayed wound model to evaluate the surrounding tissue of a wound, (its periwound) and its tissue responses following treatment with the NerveStim™ Neuropathy System, a combination topical gel and neuromuscular electrical stimulation platform. Periwound tissue was harvested at Day 14 and analyzed using NanoString gene expression profiling. Treated animals demonstrated visibly increased periwound tissue thickness compared to untreated controls. Differential expression analysis identified 76 significantly upregulated and 17 downregulated genes. Upregulated pathways included angiogenesis (Vegfa, Fgf2, Pdgfb, Nos3), neurotrophic signaling (Ngf, Bdnf, Scn9a, Trpv1), macrophage polarization (Arg1, Mrc1, Il10), and extracellular matrix remodeling (Col1a1, Col3a1, Mmp9, Timp1). Downregulation of select pro-inflammatory mediators (Nos2, Mif) was observed. These coordinated transcriptional changes are consistent with activation of reparative immune, neurovascular, and matrix remodeling pathways in diabetic periwound tissue.
Author Contributions
Academic Editor: Anubha Bajaj, Consultant Histopathologist, A.B. Diagnostics, Delhi, India
Checked for plagiarism: Yes
Review by: Single-blind
Copyright © 2026 Margaret Kalmeta, et al.
 This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Competing interests
The author declares no conflict of interest.
Citation:
Introduction
Diabetic wounds exhibit impaired healing secondary to the underlying condition of the wound’s surrounding tissue such as peripheral neuropathy, microvascular dysfunction, chronic inflammation, and extracellular matrix instability 1, 2, 3. Animal models of diabetic delayed wound healing replicate key features of human disease, including reduced angiogenesis, altered inflammatory signaling, and impaired neurovascular repair 4, 5.
Periwound tissue serves as a biologically active interface between viable tissue and the wound bed and plays a critical regulatory role in wound progression and closure 2, 6. Disruption of neurovascular signaling within this region contributes to chronicity and recurrence.
The NerveStim™ Neuropathy System combines a bioactive gel with a neuromuscular electrical stimulation device designed to target periwound tissue biology. This preclinical investigation evaluated morphological and transcriptional changes in periwound tissue in a diabetic mouse delayed wound model.
Methods
Study Design
A controlled preclinical study was conducted using a diabetic mouse delayed wound healing model to study the periwound and its release of biomarkers post treatment. Animals were assigned to:
· Treatment group: Hemastyl™ Gel + NeuroMuscular Electrical Stimulation Device
· Control group: Standard dressing alone
Periwound tissue was harvested at Day 14 following treatment initiation.
Morphological Assessment
Gross morphological images were obtained at Day 14 to assess periwound tissue characteristics.
Gene Expression Profiling
Periwound tissue underwent NanoString gene expression analysis using a custom wound healing and nerve repair codeset. Differential expression analysis was performed using predefined statistical thresholds. Genes were grouped into functional domains including inflammatory signaling, angiogenesis, neurotrophic signaling, macrophage polarization, and extracellular matrix remodeling.
Results
Periwound Tissue Morphology
By Day 14, treated animals demonstrated visibly increased periwound tissue thickness compared to untreated controls. Quantitative histomorphometry was not performed in this early-stage study.
Global Differential Gene Expression
NanoString analysis identified 76 significantly upregulated and 17 significantly downregulated genes (Figure 1). Pathway analysis demonstrated enrichment in cytokine–chemokine receptor interaction and TNF signaling pathways.
Figure 1.Volcano plot demonstrating statistically significant upregulation and downregulation of genes in periwound tissue at Day 14 following treatment with the NerveStim™ Neuropathy System, as assessed by NanoString gene expression profiling.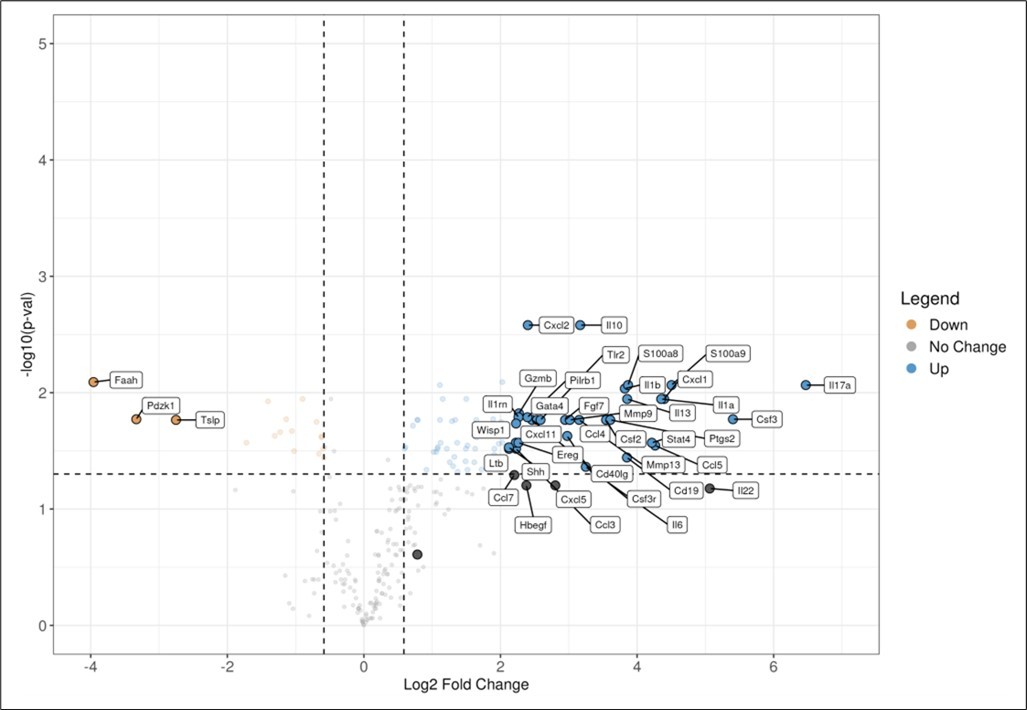
Angiogenesis and Endothelial Activation
Significant upregulation was observed in key angiogenic mediators including Vegfa, Fgf2, Pdgfb, Nos3, and Pecam1. VEGF and FGF2 are established regulators of endothelial proliferation and capillary formation during wound repair 7, 8, 9. eNOS (Nos3) supports microvascular perfusion and nitric oxide–mediated vasodilation 10.
Neurotrophic and Neural Signaling
Macrophage Polarization and Immune Regulation
Discussion
Treatment of the periwound with the NerveStim™ Neuropathy System in a diabetic mouse delayed wound model was associated with coordinated transcriptional modulation across angiogenic, neurotrophic, immune, and extracellular matrix pathways.
Hemastyl™ induces a coordinated molecular response characterized by controlled early inflammation followed by transition to a reparative phenotype. Downregulation of chronic inflammatory mediators (IL17A, MIF, NOS2), alongside increased anti-inflammatory signaling and M2 macrophage markers, supports resolution of inflammation. Concurrent upregulation of angiogenic, extracellular matrix, and neurotrophic factors is consistent with enhanced tissue repair, neovascularization, and structural remodeling, indicating effective progression through key phases of wound healing.
Upregulation of Vegfa, Fgf2, Nos3, and Pecam1 supports activation of angiogenic programs.
Concurrent elevation of Ngf and Bdnf indicates engagement of neurotrophic signaling.
Increased Arg1, Mrc1, and Il10 with reduced Nos2 expression suggests macrophage polarization toward a reparative phenotype.
Because functional nerve conduction testing, capillary density quantification, and histomorphometric analysis were not performed, conclusions are limited to gene expression and gross morphological observations.
Conclusion
In a diabetic mouse delayed wound model, treatment of the periwound with the NerveStim™ Neuropathy System was associated with transcriptional activation of angiogenic, neurotrophic, immune-regulatory, and extracellular matrix remodeling pathways within periwound tissue. These findings provide mechanistic insight into biological processes involved in healing diabetic tissue surrounding a wound that may support enhanced wound repair in diabetic environments.
Ethics Statement
All animal procedures were conducted in accordance with institutional preclinical research standards.
Funding
No external funding was received.
References
- 1.Armstrong D G, AJM Boulton, Bus S A. (2017) Diabetic foot ulcers and their recurrence. , N Engl 376-2367.
- 2.McDermott K, Fang M, AJM Boulton, Selvin E, Hicks C W. (2023) Etiology and epidemiology of diabetic foot ulcers. Clin Diabetes Endocrinol.
- 5.Galiano R D. (2004) Quantitative murine model of excisional wound healing. Wound Repair Regen. 12, 485-492.
